One Health: The health of humans and animals belongs together
To keep animals healthy, medicines are also specially developed for them. One current example is an active substance for the oral treatment of feline infectious peritonitis (FIP), a deadly disease that affects cats. New medicines are constantly supplementing the range of active substances used for treating animals, and their areas of application are becoming ever more varied. They include preparations that have been available for several years now to treat allergies. During the COVID-19 pandemic, vaccines were developed for animals. They were used on mink farms to avoid mass culls and also to protect endangered zoo animals. The innovations that result from the development of new drugs therefore benefit both humans and animals.
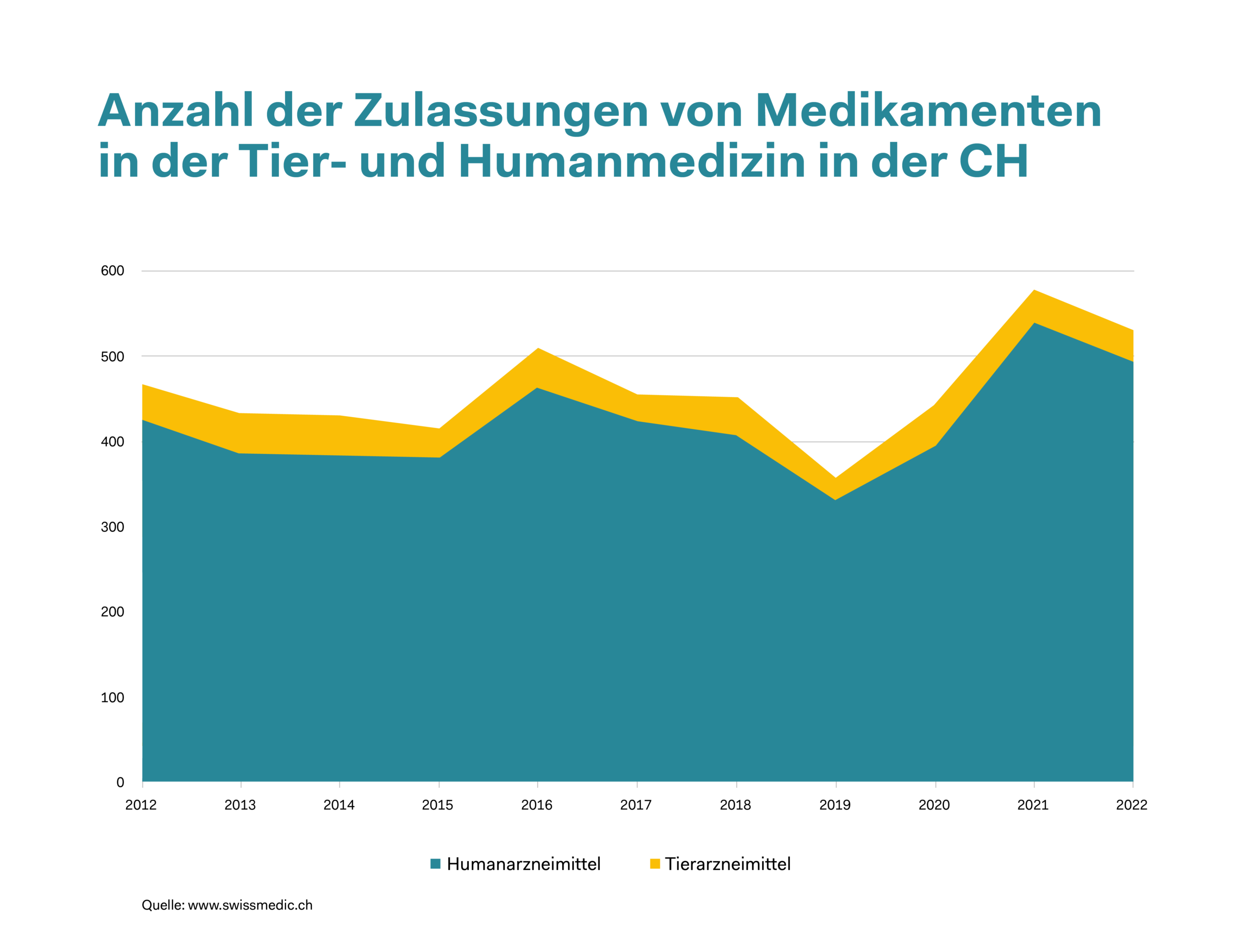
The fact that veterinary medicine is constantly benefiting from innovative drugs is illustrated by the latest approvals granted by Swissmedic. Since last year, three veterinary drugs containing a newly developed active substance have become available. Two of these substances are used in dogs – to treat painful joint inflammation and epileptic fits. The third substance is intended for use in livestock. It is an antibiotic for cows suffering from a bacterial inflammation of the udder. Drugs developed specifically for animals are also inspected prior to approval in terms of their efficacy, safety and tolerability. One way in which this is done is by clinical animal studies where one group of animals is given the new substance and a control group is given a placebo. Thanks to these studies, side effects, interactions with other drugs and dosage guidelines can be determined.
Drug approval in human and animal medicine – 2012 to 2022
Veterinary drugs are subject to strict official approval, and their quality, efficacy and safety are assessed. The safety assessment includes safety in the target animals, for users and for the environment. In livestock, the safety of the foodstuffs produced is also assessed.