One Health: The health of humans and animals belongs together
«One Health» focuses on measures for monitoring health, as well as preventing and treating diseases and injuries. The use of medicinal active substances in animals plays an important role here. These substances include vaccines, antibiotics and other drugs for the prevention and treatment of infectious diseases, as well as drugs for treating parasite infections, inflammation and pain. The diverse nature of these substances is illustrated by what are known as the “big five” – loop diuretics, pimobendan, ACE inhibitors, spironolactone and amlodipine – that are used to treat heart conditions in dogs and cats. In Switzerland, livestock and domestic animals benefit from around 700 products.
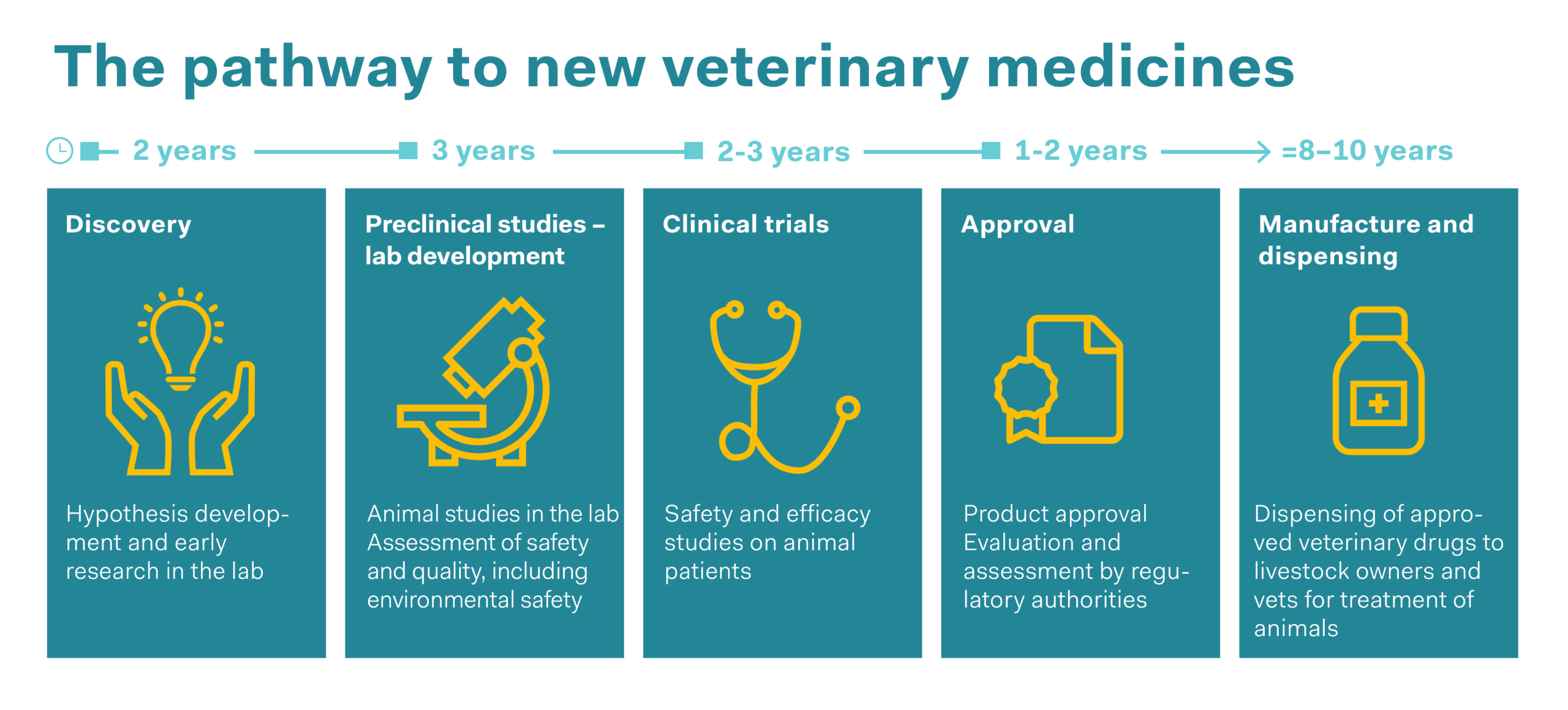
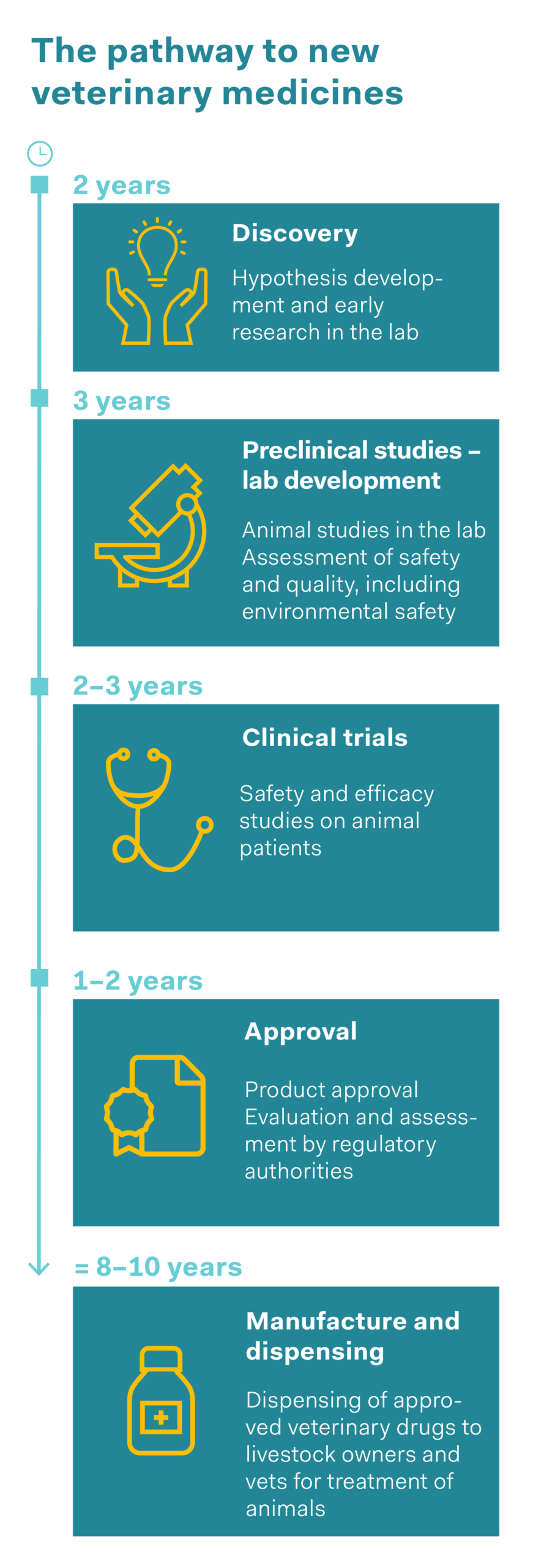
Veterinary medicines are approved in the same way as human medicines, after they have been assessed by the Swiss regulatory authority Swissmedic. This applies to medicinal products prescribed by vets as well as those that are available over the counter. Veterinary drugs are often medicines that have previously been used in humans and have then been adapted for use in animals. This means animals too benefit from the time-consuming process of drug development, from basic research and preclinical research (laboratory and animal experiments) through to clinical trials in humans. The development process, which takes an average of 12 years for a new active substance, ensures that medicines are safe and effective. Animal experiments are vital for this purpose. The 3Rs principle (replace, reduce and refine) is applied to protect the animals. Animal experiments are only ethically justifiable if no other methods are available, and the number of animals and the stress to which they are subjected are kept to an absolute minimum.